 From the perception of solid matter, it was presumed that this positive residual matter filled the entire space of the atom, giving rise to what has been called the " Thomson model" of the atom. By balancing the effect of a magnetic field on a. 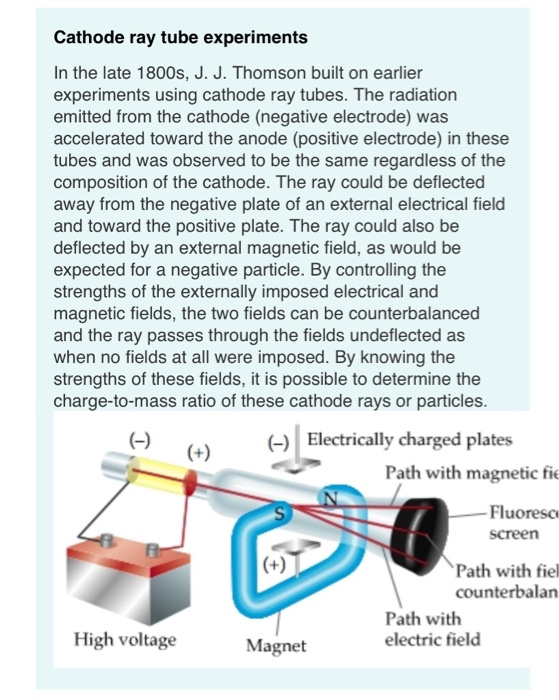
It was known that electrons could be removed from atoms, and that they became positive ions as a result. Thomson found that the cathode rays can be deflected by an electric field, as shown below. If a laboratory has only CRO in it, other measuring instruments may not be required. With the combined results of Thomson and Millikan, a value for the electron mass was obtained - a value far below that of atoms. The cathode ray oscilloscope is a versatile laboratory instrument. The determination of the charge of the electron awaited the work of Millikan who measured the electron charge in 1909 with his oil drop experiment. He also determined that the charge to mass ratio. Using this apparatus, Thomson determined the charge-to-mass ratio of the electron, e/m. Procedure of the experiment Explanation Conclusion Frequently Asked Questions FAQs Apparatus of the experiment. Thomsons experiment revealed that cathode rays are composed of negatively charged particles called electrons. The phosphors spark, or emit light, when impacted by the cathode ray. Thomson whose first experiment was to build a cathode ray tube with a. This same principle is presently used in velocity selectors for mass spectrometers. The ray can be detected by painting a material known as phosphors onto the far end of the tube beyond the anode. These rays were called Cathode Rays and were defined later as electrons. Thomson had shown that cathode rays behave as one would expect negatively charged material particles to behave. 
Thomson showed that with the application of both electric and magnetic fields, he could balance the deflections and obtain a straight beam. A narrow luminous beam could be produced by using an aperture near the cathode, and this beam could be deflected by either an electric field or a magnetic field. In the 1890's, cathode ray tubes had been developed in which a luminous beam could be produced in a partially evacuated glass tube, directed from the negative electrode (cathode) to the positive (anode). Sir Joseph John Thomson (1856-1940) played a pivotal role in developing our understanding of the electron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
